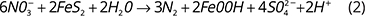|
REVIEW ARTICLE | SHORT  |
Denitrification in carbonate aquifers: a literature review
Abstract
Denitrification is the most important process for nitrate removal in groundwater. Although carbonate aquifers are important for drinking water supply, the denitrification process in these systems is less understood than for unconsolidated sedimentary aquifers. We reviewed studies that (1) provide evidence for denitrification in carbonate aquifers, (2) discuss the specific location where it occurs, and (3) quantify denitrification rates. Literature sources were identified by a systematic search of scientific databases and follow-up reference tracking.
The key finding is that denitrification in carbonate aquifers is possible, supported by microbiologic and/or isotopic evidence for several locations. Pore size and connectivity are major limiting factors for denitrification within the matrix. Denitrification in fissures/fractures would be limited by electron donor availability and unfavourable redox conditions. However, it has also been hypothesised that denitrification could occur in micro-anaerobic environments and biofilms. Denitrification rates for carbonate aquifers varied by several orders of magnitude (0.01–36 792 mg N/L/y). High concentrations and very reactive organic carbon from waste-water contamination and high groundwater temperatures are characteristic for some of the studies. Thus, rates may not be transferable to cooler oligotrophic conditions. Future research is needed to fill identified knowledge gaps.
Citation: Voutchkova et al. 2026: GEUS Bulletin 62. 8393. https://doi.org/10.34194/37b5cj23
Copyright: GEUS Bulletin (eISSN: 2597-2154) is an open access, peer-reviewed journal published by the Geological Survey of Denmark and Greenland (GEUS). This article is distributed under a CC-BY 4.0 licence, permitting free redistribution, and reproduction for any purpose, even commercial, provided proper citation of the original work. Author(s) retain copyright.
Received: 20 May 2025; Re-submitted: 20 Oct 2025; Accepted: 26 Jan 2026; Published: 30 Mar 2026
Competing interests and funding: The authors declare no competing interests.
Financial support for conducting this study was provided by the Danish Ministry of Environment.
*Correspondence: dv@geus.dk
Keywords: denitrification, denitrification rate, carbonate aquifers, chalk, limestone
Abbreviations:
ABT: acetylene blockage technique
e–: electron
OC: organic carbon OC
Edited by: Rasmus R Frederiksen (Aarhus University, Denmark)
Reviewed by: Jennifer Bearcock (British Geological Survey, UK), Rasmus J. Petersen (Aarhus University, Denmark)
1 Introduction
The term denitrification was introduced by the French scientists Gayon and Dupetit in 1882 (Payne 1986) for the microbially mediated stepwise process reducing nitrate to nitrous oxide or nitrogen gas. It is a well-studied process in water and waste-water treatment (Hiscock et al. 1991), soils, surface water-sediment interfaces (Korom 1992) and in wetlands and riparian zones (Walton et al. 2020). Denitrification in unconsolidated porous aquifers has also been studied extensively (e.g. by Postma et al. 1991; Böhlke & Denver 1995; Tesoriero et al. 2000; Kim et al. 2021; Hansen et al. 2024), as it is considered the main process of nitrate removal in groundwater (Rivett et al. 2008). However, denitrification in carbonate aquifers has received relatively little attention. This research disparity could be due to the hydrogeological complexity and geochemical heterogeneity of carbonate aquifers and subsequent methodological limitations (Section 1.2). However, carbonate aquifers are important for drinking water supply in many areas of northern Europe (West et al. 2023). For instance, they supply about one third of all drinking water in Denmark (Nilsson & Gravesen 2018; Nilsson et al. 2023), but little is known about denitrification processes in them.
Therefore, our aim was to provide an overview of the existing international peer-reviewed literature on denitrification in carbonate aquifers. We focused on studies of carbonate aquifers that (1) provide evidence for denitrification (Section 2), (2) discuss the specific location of denitrification occurrence within the aquifer (Section 3) or (3) quantify denitrification rates (Section 4). The methodology, including the search strategy, is provided in the Supplementary File.
1.1 Denitrification process
The following conditions are needed for denitrification to occur in an aquifer (Hiscock et al. 1991; Korom 1992; Rivett et al. 2008):
- Nitrate (or NO2, NO, N2O) available as electron (e–) acceptor.
- Presence of denitrifying bacteria (denitrifiers).
- Suitable e– donors, for example, organic carbon (OC) or Mn(2), Fe(2), S(–2).
- Anaerobic conditions or restricted oxygen availability (<0.2 mg/L).
- Favourable environmental conditions, for example, temperature (optimum: 25–35°C, but possible within 2–50°C), pH (5.5–8.0), availability of other nutrients (C, P, S, B, Cu, Fe, Mn, Mo, Zn and Co), and non-toxic concentrations of trace elements and anthropogenic pollutants (see Supplementary File).
The critical limiting factors for denitrification are the presence of denitrifiers, anaerobic conditions and suitable electron donor(s).
Denitrifiers are heterotrophic or autotrophic. Heterotrophs require OC for growth and maintenance, while autotrophs use reduced Mn, Fe, or S (Korom 1992). Consequently, denitrification is an autotrophic or heterotrophic process. Heterotrophic denitrification is the process of organic matter oxidation, resulting in nitrate decrease and 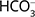 increase (Eq. 1, Otero et al. 2009). An increase in the nitrogen isotope ratio of nitrate (
increase (Eq. 1, Otero et al. 2009). An increase in the nitrogen isotope ratio of nitrate (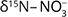 ) and stable isotope ratio of oxygen in nitrate (
) and stable isotope ratio of oxygen in nitrate (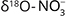 ), coupled with a decrease in the stable isotope ratio of carbon in dissolved inorganic carbon (δ13CDIC), should also be observed (Otero et al. 2009).
), coupled with a decrease in the stable isotope ratio of carbon in dissolved inorganic carbon (δ13CDIC), should also be observed (Otero et al. 2009).
Autotrophic denitrification (e.g. by pyrite oxidation) overall takes place according to Eq. 2, where the oxidation of the Fe(II) may be incomplete. Here, denitrification by pyrite oxidation results in an increase in 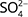 coupled with a decrease in
coupled with a decrease in 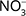 , and isotopically, an increase in
, and isotopically, an increase in 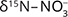 and
and 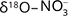 , while
, while 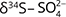 and
and 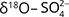 should match with the isotopic composition of
should match with the isotopic composition of 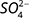 derived from S2– oxidation (Otero et al. 2009).
derived from S2– oxidation (Otero et al. 2009).
Heterotrophic denitrification is widespread across many different phyla, but autotrophic denitrification is restricted to a few closely related groups within the Proteobacteria (Gregory et al. 2014).
1.2 Hydrogeological complexity of carbonate aquifers
Most carbonate rocks, including chalk, are considered potential karst aquifers due to karstification over geological time (Bakalowicz 2005). Processes such as freeze–thaw weathering during glaciation and tectonic uplift contribute to the development of karst networks and increased transmissivity (Bakalowicz 2005; West et al. 2023).
Karstification involves the dissolution of carbonate rocks by acidic water containing dissolved CO2 (and other acids from atmospheric deposition), forming underground voids that evolve into organised, permeable structures (Bakalowicz 2005; Foley & Worthington 2023; West et al. 2023). This process requires both a solvent and groundwater flow and can occur rapidly in geological terms—within less than 50 000 years (Bakalowicz 2005). Foley and Worthington (2023) argue that all unconfined chalk aquifers should be considered karstic, if the definition of ‘karstic’ is permeability, enhanced by dissolution.
Chalk aquifers are recognised as karstic but differ from other carbonate aquifers due to higher primary porosity, fracture frequency, and mechanical weakness (West et al. 2023). Porosity in chalk includes matrix, fracture and fissure types, each influencing water storage and solute transport (Foley & Worthington 2023). Fissures may form by dissolution of existing fractures or at lithostratigraphic boundaries due to concentrated flow. Solutional porosity significantly affects aquifer behaviour, and changes in groundwater flux can alter karst structures (Bakalowicz 2005; West et al. 2023). However, permeability contrasts between matrix and conduits may limit or prevent groundwater exchange (Bakalowicz 2005).
Carbonate aquifers exist along a spectrum from purely fractured to fully karstic systems (Bakalowicz 2005). In Denmark, carbonate aquifers (often covered by Quaternary sediments) have been underrecognised as karstic, despite evidence of local karst features, especially where sediment cover is less than 15–20 meters (Nilsson et al. 2023).
Karstic characteristics significantly influence the conceptualisation and numerical modelling of aquifers yet are often overlooked (West et al. 2023). Classical methods such as borehole observations, pumping tests and distributed models are generally ineffective in karst aquifers due to their inability to represent the complex heterogeneity (Bakalowicz 2005). Observations from boreholes lack representativity for the aquifer, and flow regimes in karst systems often deviate from Darcy flow, exhibiting turbulent and conduit-dominated hydraulics (Bakalowicz 2005).
The dissolution-driven development of a hierarchical network of connected fissures results in non-random permeability distribution (Foley & Worthington 2023). Karst aquifers are inherently heterogeneous and anisotropic, with unique structural and hydraulic characteristics that challenge generalised modelling approaches. Therefore, they must be studied on a case-by-case basis to account for site-specific complexities (Kalhor et al. 2019).
2 Evidence for denitrification in carbonate aquifers
Microbial or isotopic evidence of denitrification in carbonate aquifers has been provided by several studies, but none for Danish carbonate aquifers. Nitrate-reducing bacteria were found down to 10 m and 50 m depth in the Middle and Upper Chalk Formations in the United Kingdom respectively (Whitelaw & Rees 1980). Middle and Upper Chalk (old) refer to the White Chalk Subgroup (new), while Lower Chalk (old) refers to the Grey Chalk Subgroup (new). Note that throughout the paper we use the terminology as it is in the cited papers. Parker and James (1985) isolated facultative aerobes (Pseudomonas) and demonstrated their nitrate-reduction potential from the anoxic Cretaceous chalk in Norfolk (UK, down to 40 m depth). Denitrifying bacteria were also cultured from samples scraped from fissure walls of the Jurassic limestone in Lincoln (UK; Hiscock et al. 1991). Gregory et al. (2014), however, stated that very little is known about the microbial communities in the Cretaceous chalk in the UK in general, compared to other UK aquifers. Le Bideau (2012) isolated Thiobacillus denitrificans from Oxfordian limestone (France). Jakus et al. (2021) enriched a novel lithoautotrophic culture from nitrate-containing groundwater of a pyrite-rich limestone aquifer in Southern Germany (Middle Triassic carbonates of the Upper Muschelkalk). The culture was dominated by Fe(II) oxidising microorganisms affiliated with the Gallionellaceae family that can perform nitrate reduction coupled to Fe(II) oxidation, leading to N2O and N2 formation without addition of organic substrates (Jakus et al. 2021).
Further evidence for denitrification in carbonate aquifers based on isotopic data was provided by Griggs et al. 2003; Otero et al. 2009; and Hernández-del Amo et al. 2018. In the oxic Osona basin limestone aquifer (Spain), Otero et al. (2009) observed a 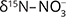 range (Supplementary File, Table S1) that could either be due to volatilisation or denitrification, but the positive correlation between
range (Supplementary File, Table S1) that could either be due to volatilisation or denitrification, but the positive correlation between 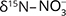 and
and 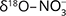 values was interpreted as evidence for denitrification. For about c. 25% of the samples there was clear evidence, and the natural attenuation was approximated up to 25–33%. The oxic environment should have been unsuitable for denitrification, so the authors hypothesised that denitrification occurs in micro-anaerobic environments. The dominating process was interpreted as autotrophic (denitrification by sulphide oxidation), based on comprehensive isotopic characterisation of the ions involved in the denitrification reactions (
values was interpreted as evidence for denitrification. For about c. 25% of the samples there was clear evidence, and the natural attenuation was approximated up to 25–33%. The oxic environment should have been unsuitable for denitrification, so the authors hypothesised that denitrification occurs in micro-anaerobic environments. The dominating process was interpreted as autotrophic (denitrification by sulphide oxidation), based on comprehensive isotopic characterisation of the ions involved in the denitrification reactions (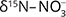 and
and 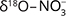 ,
, 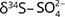 ,
, 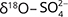 , and δ13C–DIC). For the same aquifer, Hernández-del Amo et al. (2018) estimated slightly lower attenuation by denitrification (<10–25%) than Otero et al. (2009). Griggs et al. (2003) confirmed microbially mediated denitrification as the primary factor for nitrate decrease of waste water injected into the Key Largo limestone (USA). The systematic variation of
, and δ13C–DIC). For the same aquifer, Hernández-del Amo et al. (2018) estimated slightly lower attenuation by denitrification (<10–25%) than Otero et al. (2009). Griggs et al. (2003) confirmed microbially mediated denitrification as the primary factor for nitrate decrease of waste water injected into the Key Largo limestone (USA). The systematic variation of 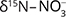 , δ15N–N2 and the isotope fractionation factor (−12 ± 4‰) provided strong evidence for denitrification in the brackish and fresh waters of the aquifer (Griggs et al. 2003). The co-injection of waste water presumably implied that the e– donor was the waste water OC, and their results showed that the carbonate rock can host active denitrifiers.
, δ15N–N2 and the isotope fractionation factor (−12 ± 4‰) provided strong evidence for denitrification in the brackish and fresh waters of the aquifer (Griggs et al. 2003). The co-injection of waste water presumably implied that the e– donor was the waste water OC, and their results showed that the carbonate rock can host active denitrifiers.
3 Location of denitrification within a carbonate aquifer
Denitrification in double (triple)-porosity systems, such as carbonates, could potentially occur in the matrix, on the walls of the fissures and fractures, or in both places (Fig. 1).
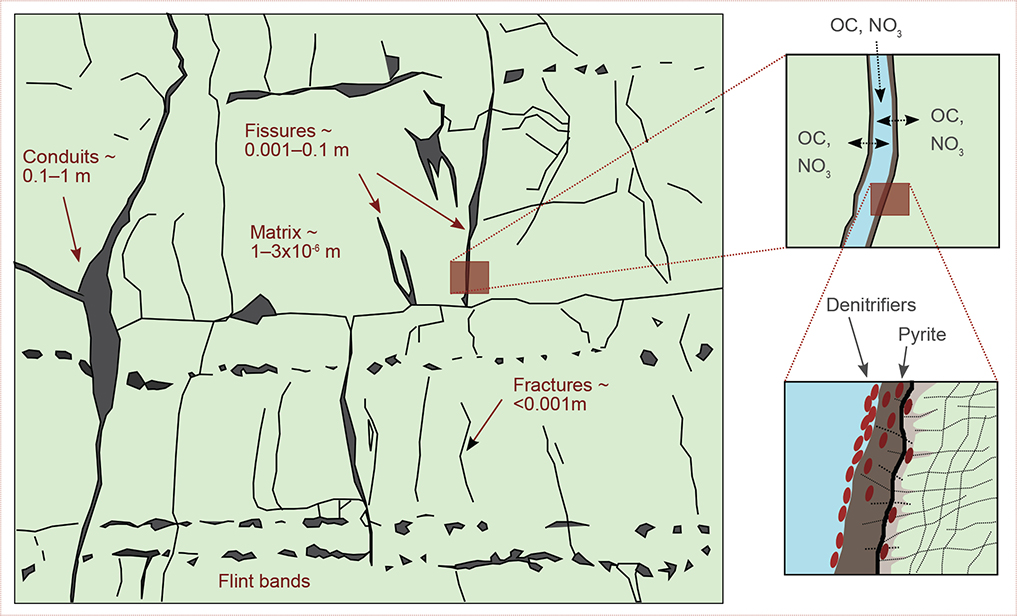
Fig. 1 Conceptual model showing a carbonate aquifer with zoom at a pyrite-coated fissure wall, where it is hypothesised that denitrifiers are found; denitrification is potentially also happening in the matrix, close to the fissure wall (see text for references). The background fracture pattern and the pore sizes are digitised from Maurice et al. (2023). OC: organic carbon. NO3: nitrate.
Pore size could be a limiting factor for microbial growth (Whitelaw & Rees 1980; West & Chilton 1997; Rivett et al. 2008; Le Bideau 2012), which has been noted for some carbonate deposits in Europe (Rivett et al. 2008). For unconsolidated aquifers, when both pores and pore throats were >1 µm the bacteria (with a diameter 1 µm) were assumed to be active and motile, but if the pore throats were smaller, bacteria would be trapped in the pore spaces (Rebata-Landa & Santamarina 2006). West and Chilton (1997) hypothesised that microbial colonisation of the matrix of northern European chalk would be difficult, because although the chalk may have high porosity (Table 1), the pore size is small (median diameter of pore throats 0.5 µm). The matrix porosity of the Oxfordian limestone (France) was also assessed to be too fine to permit colonisation (Table 1, Le Bideau 2012). Danish carbonate aquifers have porosities in the range 10–49 %, but the pore size could be a limiting factor as the peak diameter is c. 0.7 µm (Table 1). Future research is needed to provide evidence for presence of denitrifiers and active denitrification in the Danish carbonate aquifers.
| Aquifer | Lithology | Matrix porosity (%) | Pore-size diameter (µm) | Fissure spacing (m) | Reference |
| Cretaceous chalk (Norfolk, UK) | Fissured microporous | 25–45 40–50 |
0.2–1.0 | 5–10 (macro-fissure), 0.1 (micro-fissure) | (West & Chilton 1997) (Parker & James 1985) |
| Jurassic limestone (Lincoln, UK) | Microporous or dense | 10–25 | 0.05–0.5 | 1–5 (macro-fissure) | |
| Middle Chalk Formation (Eastbourne, UK) | 37–42 | 0.53 ± 0.14 | n.a. | (Whitelaw & Rees 1980) | |
| Upper Chalk Formation (Winchester, UK) | Uniform fine-grained | 0.65 ± 0.14 | n.a. | ||
| Oxfordian limestone (Poitou-Charentes, Fr) | Fractured | n.a. | <1 | n.a. | (Le Bideau 2012) |
| Middle Triassic carbonates (Upper Muchelkalk in South-West Germany) | Back shoal facies (coastal lagoonal setting) | 15–28 | <1–280 | n.a.; fractured and karstified | (Osenbrück et al. 2022) |
| Shoal facies | 15–30 | <1–1700 | |||
| Tempestite facies (storm deposits of the mid to deeper ramp) | 0.5–10 | <1–1500 | |||
| Basinal facies (basinal setting below storm wave base) | 0.5–5 | n.a. | |||
| Upper Cretaceous chalk (Skrivekridt, DK) | Fine-grained, white micrite chalk, occasionally chert and thin marl layers | 20–50 | n.a. | n.a. | (Kidmose et al. 2023) |
| Middle Danian limestone (Bryozokalk, DK) | Bryozoan reef limestone | 5–45 | n.a. | n.a. | |
| Late Danian limestone (Kalksandskalk, DK) | Calcarenite, with thick chert layers | 10–40 | n.a. | n.a. | |
| Upper Cretaceous chalk (outcrop Stevns Klint, DK) | Heterogeneous | 48.8 | 0.022–1.46 1 | n.a. | (Khan et al. 2023) |
| Upper Cretaceous chalk (outcrop Rørdal quarry, Aalborg, DK) | Heterogeneous | 47.0 | 0.020–1.24 2 | n.a. | |
| Note that throughout the present paper we have kept the terminology as it is in the cited papers. Middle and Upper Chalk (old) refer to the White Chalk Subgroup (new), while Lower Chalk (old) refers to the Grey Chalk Subgroup (new). n.a.: not available in the reference. 1Peak diameter of the pore-size distribution: 0.68 µm. 2Peak diameter of the pore-size distribution: 0.74 µm. |
|||||
Already in the 1980s, it was hypothesised that denitrification in the Middle and Upper Chalk Formations (UK, Table 1) would be limited to the fissures, where denitrifiers would adhere to the fissure walls and derive their nutrients from the diffusion of pore waters (Whitelaw & Rees 1980). The presence of bacteria down to 50 m suggested fissure continuity, while high bacterial counts indicated that fissure walls were favourable sites for bacterial growth and activity (Whitelaw & Rees 1980). The authors hypothesised that bacteria populations have existed within the rock formation and/or migrated from the ground surface to the chalk. Because of low nitrate in the pore water, either a dynamic nitrate production and utilisation resulted in a low steady-state concentration, or bacteria survived in a non-growing phase. Parker and James (1985) hypothesised that the microaerobic conditions (presumably needed by the Pseudomonas) were likely to be present in fissures. Although more recently it has been shown that denitrification by specific strains of Pseudomonas can be performed in both microaerobic and anaerobic conditions with similar efficiency (Wang et al. 2025). Biofilms were hypothesised as a possible and favourable location for denitrification within an oxic karstified aquifer (Henson et al. 2017).
Hiscock et al. (1991) suspected heterotrophic denitrification and hypothesised that the e– donor (OC) must be within the matrix (Supplementary File, Table S2), however the very small pore size (Table 1) would limit the denitrification to short distances from the fissure walls of the Jurassic limestone in Lincoln (UK, Table 1). Similarly, with respect to the Cretaceous chalk in Norfolk (UK, Table 1), OC in the mobile fissure-water was insufficient to support significant denitrification in contrast to pore water which contained higher concentrations (Supplementary File, Table S2). Parker and James (1985) further hypothesised that the pore-water OC was derived from the rock matrix, as it could contain >1000 mg total OC per kg dry weight. On the other hand, Le Bideau (2012) hypothesised that denitrifiers live attached to the pyritic walls of fractures and filter the mobile fissure water in the Oxfordian limestone (France). Their petrographic analysis showed that pyrite was indeed present in both the matrix and the fissures. Osenbrück et al. (2022) studied the nitrate reduction potential of the Middle Triassic carbonates of the Upper Muschelkalk (Germany) where denitrification was also due to Fe(II) and pyrite oxidation (pyrite concentrations were up to 4% by weight). They concluded that the high-porosity carbonate rock facies (Table 1), together with hydraulically active, pyrite-coated fractures, were the zones of highest nitrate reduction potential within the aquifer (Osenbrück et al. 2022). The nitrate reduction potential increased with increasing porosity of the rock matrix and decreased with increasing hydraulic conductivity (or effective fracture aperture) and spacing of the fracture network (Osenbrück et al. 2022). Einsiedl and Mayer (2006) hypothesised that some denitrification had occurred in the porous rock matrix of the Upper Jurassic karst aquifer (Southern Germany), where the rate-limiting step was most probably the diffusion of nitrate from the preferential flow paths in the fractured aquifer to the more immobile matrix water.
4 Denitrification rates in carbonate aquifers
To the best of our knowledge, denitrification rates (or just ‘rates’) for Danish carbonate aquifers have not been reported in the peer-reviewed literature. To facilitate intercomparison between the studies reviewed here, we converted all zero-order rates to mg N/L/y (the reported units were μmol/m3gw/h, µmol N/kg/d, mmol/L/h, µg N/kg/d, µmol N/L/y, nmol N2O/g/d, µmol N/L/d) and then transformed them to logarithm (loge) values (Kim et al. 2021). The rates for carbonate aquifers varied by several orders of magnitude: 0.01–36 792 mg N/L/y, or as loge(mg N/L/y) –4.6–+10.5. Both the highest and lowest rates were estimated for the same aquifer in Silver Springs (Florida (FL), USA, Yang et al. 2023). First-order rates (by Griggs et al. 2003 and Katz et al. 2012) were converted to /y.
Although we converted all units, the variety of methods used (Table 2) makes direct comparison challenging as rates are estimated at different scales and conditions (e.g. in situ vs. lab, large scale vs. point measurement). Groffman et al. (2006) reviewed the diversity of methods for quantifying denitrification and discussed their constraints related to scale (time and space) representation. Henson et al. (2017) also pointed out that the disparity among rates, derived from different methods, illustrates the general challenge of upscaling rates determined at the local scale (point measurements) to the regional scale. Kim et al. (2021) showed also that age vs. N2 mass balance method estimated rates in the lower range, the tracer methods in mid-range, while the lab experiments resulted in the highest rates. This was due to scale and method specifics (Kim et al. 2021): the age versus mass balance method represents the average rate of the entire recharge area of the sampling point (lower-range rates); the tracer tests focus on the local conditions and are potentially biased to locations where it is possible to measure (relatively shallow and near the streams); while the laboratory methods measure the highest rates, potentially because of sample mixing, that is, the activation of reaction surfaces and in many cases more favourable conditions. Methods for upscaling from the point–scale measurements by acetylene blockage technique (ABT) to catchment scale was presented by Kim et al. (2021) for a glacial aquifer in Denmark. ABT is one of the most popular methods for measuring potential denitrification in soil and unconsolidated sediments, however here only Morris et al. (1988) used it. Another concept for assessing denitrification by comparing nitrate trends in oxic and anoxic nitrate-reducing zones at local and national scales was introduced recently (Hansen et al. 2025).
| Location | Country (State) | Carbonate aquifer | Method | Depth | Ratea | Note | Ref. | |
| m | mg N/L/y | loge(mg N/L/y) | ||||||
| Florida Keys | USA (FL) | Pleistocene limestone | tracer test | 4.5–18 | 332–898 | 5.8–6.8 | Waste disposal site | (Corbett et al. 2000) |
| Key Colony Beach | USA (FL) | Pleistocene limestone | excess-N2, travel times | 16.8–18.3 | 0 | -5 | SF6 tracer travel times: 10 d (B-18) | (Griggs et al. 2003) |
| 7.6–9.1 | 38.4 | 3.6 | SF6 tracer travel times: 7 d (C-9) | |||||
| 11.5 | 2.4 | SF6 tracer travel times: 60 d (A-9) | ||||||
| 22.7 | 3.1 | SF6 tracer travel times: 55 d (D-9) | ||||||
| 2.92b | First order rate, unit /y | |||||||
| Ichetucknee Springs | USA (FL) | Paleogene limestone, unconfined | tracer test | 6–15 | 3682 | 8.2 | Oxic site (MW14), injected Cl, NO3 | (Henson et al. 2017) |
| 6136 | 8.7 | Oxic site (MW14), injected Cl, NO3, Fumarate | ||||||
| 8–16 | 3682 | 8.2 | Anoxic site (MW15), injected Cl, NO3 | |||||
| 15 955 | 9.7 | Anoxic site (MW15) injected Cl, NO3, Fumarate | ||||||
| Oak Park | Ireland | Quaternary gravel and sand/limestone | tracer test with 15N | 13–15 | 1.8 | 0.6 | (Jahangir et al. 2013) | |
| Limestone | 20–22 | 0.4 | -1.0 | |||||
| Middle Claiborne aquifer | USA (TN/MS) | Eocene limestone, sand, clay, silty, confined | nitrate + age | 41.8–160 | 0.07–0.14 | -2.7 – -2.0 | (Katz et al. 2012) | |
| 0.08–0.16b | First order rate, unit /y | |||||||
| Parris Island | USA (SC) | Miocene/Oligocene bryozoan limestone (biosparite) | ABT | 32 | 1.8 | 0.6 | depth-specificc | (Morris et al. 1988) |
| 47 | 3.5 | 1.3 | depth-specificc | |||||
| Pleistocene sand/silt and weakly cemented bryozoan limestone | 5–185 | 5.4 | 1.7 | depth-average (mix of lithologies), 8 depths | ||||
| Silver Springs | USA (FL) | Paleogene limestone and dolomite | tracer test | 41–44 | 36 792 | 10.5 | matrix (M-0779) | (Yang et al. 2023) |
| 18–21 | 27 083 | 10.2 | matrix (M-0780) | |||||
| 8–11 | 0 | -5 | matrix (M-0781) | |||||
| 34–37 | 0 | -5 | non-matrix, maybe limited by short residence times 3h (M-0789) | |||||
| 18–20 | 0 | -5 | non-matrix, maybe limited by short residence times 3h (M-0827) | |||||
| 27–30 | 0 | -5 | matrix (M-0789) | |||||
| 24–27 | 0 | -5 | matrix (M-0827) | |||||
| mass-balance N2, residence time | 26–28.5 | 6.6 | 1.9 | non-matrix, whole aquifer | ||||
| 24.3–27.1 | 0.05 | -3.0 | all together, whole aquifer, average | |||||
| 23.6–26.6 | 0.01 | -4.6 | matrix, whole aquifer | |||||
| Lincolnshire | UK | Fissured and confined limestone | nitrate gradient along flow-paths | n.a. | 3.65–18.25 | 1.29–2.9 | (Hiscock et al. 1991) | |
| Poitou-Charentes | France | Oxfordian limestone | nitrate gradient and effective velocity | n.a. | 120.5 | 4.8 | (Le Bideau 2012) | |
| Salento | Italy | Cretaceous limestone aquifer | Lab experiments (see Supplementary File for details) | n.a. | 110 | 4.7 | low DO conditions (DO <0.3 mg/L) | (Masciopinto 2007) |
| 201 | 5.3 | oxic conditions (DO = 0.9) and low OC | ||||||
| 766.5 | 6.6 | oxic conditions and high OC | ||||||
| ABT: acetylene blockage technique; n.a.: not available. With bold: first order rates with unit y-1. aThe rounding of the rates was done after unit conversion and loge- transformation, for visualisation purposes. bFirst order rate, the unit here is y-1. cThe results were digitised from fig. 3 of Morris et al. (1988). |
||||||||
The rates from Corbett et al. (2000), Griggs et al. (2003) and Morris et al. (1988) most probably reflect high and very reactive OC conditions, as the studies focused on groundwater contaminated by waste water.
The groundwater temperatures of three carbonate aquifers were in the range 18.4–25.3°C (Griggs et al. 2003; Katz et al. 2012; Yang et al. 2023). These temperatures are very high in comparison to North European conditions (e.g. typical groundwater temperature in Denmark is 8–9°C), thus lower rates could be expected under cooler climatic conditions. Masciopinto (2007) conducted their laboratory experiments at 15°C (Supplementary File), and the rest of the studies did not report temperature.
There were methodological limitations in several studies, most probably due to aquifer heterogeneity. In several studies, quantification of rates was limited to specific tests only. For instance, Griggs et al. (2003) were unable to calculate rates from a 10-day travel time tracer test in the Key Colony limestone (FL, USA). However, they succeeded for other depths and travel times within the same aquifer. At Silver Springs limestone and dolomite (FL, USA), rates could not be quantified for matrix or for non-matrix flow, based on tracer tests in five different places/depths (Yang et al. 2023). Yang et al. (2023) explained it with the short residence times for non-matrix flow locations (3h). However, they also found hotspots in the same aquifer where the highest rates across all studies included here were 27 083 and 36 792 mg N/L/y (loge(mg N/L/y) = 10.2 and 10.5, respectively). Two studies on chalk aquifers in the UK also failed in quantifying rates: Parker and James (1985) stated that it was “virtually impossible” to calculate in situ denitrification for the Norfolk Chalk aquifer, while Howard (1985) could not calculate rates for the fine-grained Late Cretaceous limestone in eastern England.
Henson et al. (2017), on the other hand, observed nitrate loss based on results from push-pull tracer tests irrespective of redox state (oxic and anoxic sites) or treatment (nitrate or nitrate + fumarate), but there was no detectable denitrification activity based on microbial rRNA expression data. They attributed the observed nitrate loss to dissimilatory nitrate reduction to NH4 (at the anoxic site with nitrate treatment) and assimilatory reduction of nitrate to NH4 (at both sites with nitrate + fumarate treatment).
5 Conclusion
Based on the limited peer-reviewed literature on denitrification in carbonate aquifers, we can conclude that denitrification in carbonate aquifers is possible, and for several locations there is isotopic or/and microbiological evidence for it. Denitrification rates in carbonate aquifers varied over several orders of magnitude: 0.01–36 792 mg N/L/y or as loge(mg N/L/y) –4.6 to 10.5. These are generally comparable with the ranges in unconsolidated sedimentary aquifers (Korom 1992; Rivett et al. 2008; Kim et al. 2021; Hansen et al. 2025). Interestingly, both the lowest and highest rates here were for the same Paleogene limestone and dolomite aquifer in Silver Springs (USA), illustrating the heterogeneity of carbonate (karstified) aquifers. It should be noted that some of the reported rates may be due to specific favourable conditions, especially high concentrations and reactivity of the organic content (waste water-polluted groundwater) and perhaps high groundwater temperatures (>20°C).
Denitrification is hypothesised to be possible both within the matrix and the fissures. However, pore size and pore connectivity could be a limiting factor for bacterial colonisation of the aquifer matrix. Thus, for aquifers with pore sizes smaller than the denitrifiers, denitrification would likely occur within or very close to the fracture and fissure walls, and it would be limited by e– donor supply and unfavourable conditions (e.g. oxic environment). It was also hypothesised that micro-anaerobic environments and biofilms within fissures and conduits would provide favourable conditions as well, but this hypothesis has not yet been tested.
Overall, further research is needed to elucidate the denitrification process for carbonate aquifers, especially for those located in northern Europe, including Denmark. There is a need for future studies on the location of denitrification within the carbonate rocks, the source, type and distribution of e– donors in the rock-matrix system, pore-water diffusion, and transport of nitrate and e– donor in the flowing groundwater within the fissures, identification of denitrifiers present in the aquifers, quantification of denitrification rates at different scales and assessing the relevance of other  attenuating processes.
attenuating processes.
Acknowledgements
This study was conducted as part of the Danish Environmental Protection Agency project “Improved nitrogen retention mapping for a new regulation model for agriculture” (2023–2024) for which Anker Lajer Højberg (GEUS) was the project’s principal investigator (PI). The authors wish to thank the reviewers for their comments, which improved the manuscript considerably.
Additional information
Author contributions
Conceptualisation: All
Investigation: DV
Methodology: DV, HK
Project Administration: BGH
Writing – original Draft: DV
Writing – Review & Editing: All
Additional files
The Supplementary File details the literature review method and is available at https://doi.org/10.22008/FK2/MD5QBY.